Packs sizes (2)
| code | packaging size | price per unit | box price per unit | |
|---|---|---|---|---|
| Code & packaging | Price per piece | |||

|
code
361954.1611
|
packaging size
1000 ml
|
price per unit
single
$284,25
|
box price per unit
$241,61x 6 units
|

|
code
361954.1612
|
packaging size
2.5 l
|
price per unit
single
$639,00
|
box price per unit
$543,15x 4 units
|
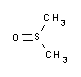
Technical data
- Melting Point:
- 18.4 °C
- Boiling Point:
- 189 °C
- Density:
- 1.103 kg/l
- Refractive Index:
- 20/D 1.477
- Physical Description:
- liquid
- Product Code:
- 361954
- Product Name:
- Dimethyl Sulfoxide for UV, IR, HPLC, GPC
- Quality Name:
- for UV, IR, HPLC, GPC
- Specifications:
- Minimum assay (G.C.): 99.9%
Maximum limit of impurities
APHA colour: 10
Acidity: 0.0005 meq/g
Alkalinity: 0.0002 meq/g
Non-volatile matter: 0.001 %
Water (H2O): 0.05 %
Suitability for IR spectrometry:: passes test
Fluorescence at 365 nm (as quinine): 7 ppb
UV Spectrum (1cm cell; Ref.: water):
Transmittance at 265 (Cut off) nm: ≥10%
Transmittance at 270 nm: ≥30%
Transmittance at 280 nm: ≥63%
Transmittance at 290 nm: ≥70%
Transmittance at 310 nm: ≥80%
Transmittance at 330 nm: ≥94%
Transmittance at 350-450 nm: ≥98%
Data of interest in HPLC:
Rohrschneider Polarity: 7.2
Eluotropic value e° (Al2O3): 0.75
Sol. H2O in solv. at 20°C: miscible
For critical jobs, purge with nitrogen.
Microfiltered product (0.2 μm) and bottled under nitrogen atmosphere.
- WGK:
- 1
- Storage:
- Storage away from direct light.
- Master Name:
- Dimethyl Sulphoxide
- Synonyms Long Text:
- DMSO, Methylsulphoxide, Sulphinylbismethane
- EINECS:
- 200-664-3
- CS:
- 29309095
Documents
Inquiry
Comments
DMSO - Production and synthesis - Technically, dimethyl sulfoxide is produced from dimethyl sulfide, for example, by catalytic oxidation with dinitrogen tetroxide in the presence of oxygen. In the laboratory, the thioether dimethyl sulfide can be oxidized with stoichiometric amounts of hydrogen peroxide or diluted nitric acid. However, the dimethyl sulfoxide can be further oxidized to dimethyl sulfone. DMSO is also a by-product of pulp production. – Properties- Physical properties - Dimethyl sulfoxide is a colorless and odorless hygroscopic liquid. After prolonged storage, it often has a foul odor (of dimethyl sulfide). With a melting point at 18 °C, the substance can solidify only slightly below room temperature. The enthalpy of fusion is 14.37 kJ·mol−1. At normal pressure, the compound boils at 189 °C. The vapor pressure curve is obtained according to Antoine corresponding to log10(P) = A−(B/(T+C)) (P in bar, T in K) with A = 4.49107, B = 1807,002 and C = −60,995 in the temperature range from 325.5 to 442.1 K, o with A = 5.23039, B = 2239.161 and C = −29.215 in the temperature range from 293 to 323. It is miscible in any ratio with water, furthermore with many organic solvents such as alcohols, carboxylic acid esters, ketones, and chlorinated hydrocarbons. It belongs to the group of nucleophilic, aprotic, dipolar solvents (such as dimethylformamide). - Chemical properties - The compound begins to decompose thermally at normal pressure at the boiling point of 189 °C, which can be violent to explosive. The decomposition is catalyzed by acids or bases, so that it can become relevant at much lower temperatures. Violent to explosive decomposition also occurs in the presence of halogen compounds, metal nitrates, metal perchlorates, sodium hydride, periodic acid and fluorinating agents. - Dimethyl sulfoxide can be deprotonated with sodium hydride or sodium amide to form a methylsulfinyl carbanion (dimsyl anion), which is used as a very strong nucleophilic reagent in organic synthesis. The reaction mixtures can decompose explosively above 70 °C. There is also a risk of explosion when isolating the solid sodium salt. - Safety parameters - Dimethyl sulfoxide forms flammable vapor-air mixtures above the flash point at 88 °C. The lower explosion limit is 1.8% v/v (58 g/m³). An upper explosion limit (UEL) cannot be determined because of the thermal decomposition of the substance. The limiting oxygen concentration was determined to be 3.9% at 200 °C. The ignition temperature is 270 °C. The substance thus is included in temperature class T3. The electrical conductivity is rather low at 2·10−7 S·m−1. - Deuterated dimethyl sulfoxide, DMSO-d6 - Fully deuterated DMSO (DMSO-d6), in which all six hydrogen atoms have been replaced by deuterium, is used as a solvent in NMR spectroscopy. – Pharmacology - Dimethyl sulfoxide has anti-inflammatory (antiphlogistic) and analgesic properties. It is therefore used therapeutically as a percutaneous (from Latin through the skin) drug for the treatment of local pain conditions (for example, in sports injuries or rheumatic conditions). Since DMSO helps to quickly reduce swelling in bruises, it is used especially in martial arts when needed. A study from Brazil was able to show that DMSO gel and therapeutic ultrasound have significantly better results with regard to the decongestion of blunt injuries compared to other forms of treatment (or no treatment). - Its special ability is the easy penetration into skin and other cell membranes. It is therefore used as a carrier substance in drugs applied to the skin (ointments, gels, plasters, tinctures) for the introduction of active ingredients such as heparin or analgesics as a so-called transport mediator (also called penetration enhancers), i.e., substances dissolved in DMSO are easily absorbed by the organism through the skin. This also applies to poisons that are otherwise no or weakly effective contact poisons, such as cyanides. Therefore, solutions of compounds considered toxic must be rinsed off with suitable means (e.g., water) immediately if they come into contact with the skin. – Uses - DMSO is a widely used solvent in laboratory and technical applications. For example, it is used in spinning solutions of polyacrylonitrile, as a paint stripper, as a solvent in aromatics extraction, and as a reaction medium in organic syntheses. Many inorganic salts also have good solubility in DMSO. - In organic synthetic chemistry, it serves as an oxidant in Swern oxidation and Parikh-Doering oxidation. In pharmacy, it serves as an ingredient in ointments (see pharmacology). - In cell culture, DMSO finds use in freezing media in the cryopreservation of eukaryotic cells. As an antifreeze, it prevents the formation of ice crystals during the freezing process; these can destroy cell organelles and thus lead to cell death. DMSO inhibits crystal formation somewhat better than glycerol. - Most substance libraries also use DMSO as a solvent. However, since the solvent is not completely inert, this can lead to a deterioration of sample purity.Freezing of cells for cell culture :Dimethyl sulfoxide (DMSO) is widely used for the freezing of cells in cell culture instead of glycerol. DMSO is added to prevent the formation of ice crystals during the freezing process, otherwise cells would be destroyed. DMSO is commonly used at the concentration of 10%. Please note: DMSO is toxic to cells! Therefore it is advisable to use pre-chilled "freeze medium". Usually, such freeze medium consists of standard culture medium supplemented with 10% DMSO. Some researchers use 90% serum supplemented with 10% DMSO as freeze medium, which enhances the viability in some cell types. Particular cell types cannot be transferred into liquid nitrogen directly, but are slowly frozen to -70°C in a refrigerator. For very slow cooling of these stocks, cells are frozen well insulated and subsequently transferred to liquid nitrogen. Thawing: For further cultivation thawing should be carried out as quickly as possible. After thawing we recommend to wash the cells in medium or to change the medium the day after plating.



